Dr. David Martyn is an Associate Professor of Organic Chemistry in the Chemistry & Physics Department at Southwestern Oklahoma State University (SWOSU).
EDUCATION
1999 – B.S. Chemistry, Southern Nazarene University
2003 – M.S. Chemistry, University of Oklahoma
2006 – Ph.D. Chemistry, University of Oklahoma
2008 – Began teaching at SWOSU
RESEARCH
Synthesis of amine functionalized cholesterol derivatives for use in metal-complex based imaging agents
Dr. David Martyn (Mentor)
Cholesterol, an essential component of cell membranes, and cholesterol derivatives, essential cell regulators, have been widely studied as anticancer agents and cancer diagnostic agents. Cholesterol-derivatization allows incorporation of pharmaceutical “hitch-hikers” into cells via the usual cellular mechanisms. Typically, this cholesterol-derivatization makes direct use of the hydroxyl group on carbon three of the cholesterol structure. In this research we will explore the possibility of modifying the existing hydroxyl group and converting the vinyl group on carbons five and six into an additional binding site. Ultimately, we envision synthesis of a metal binding, cholesterol-derived tetraazamacrocycle as seen in the figure below.
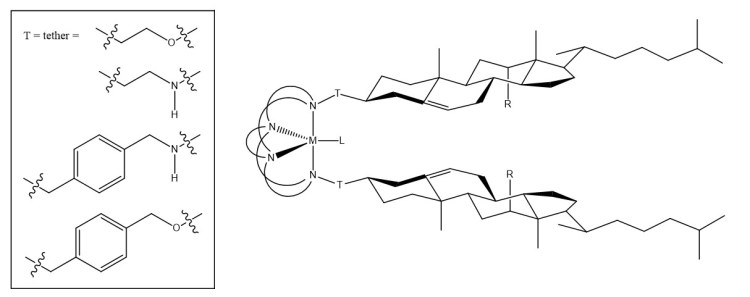
In this project, we seek to develop a metal complex-based cholesterol-derived tetraazamacrocycle for use in diagnostic imaging. The first step of this process involves synthesizing amine-substituted cholesterol derivatives with cholesterol serving as the starting material. Conversion of the carbon three hydroxy group into ether and amino groups will allow synthesis of starting materials capable of being introduced and incorporated during synthesis of the tetraazamacrocycle. Additionally, through hydroboration‐oxidation, (3β)‐cholest‐5‐en‐3‐ol (cholesterol) will be converted into (3β,5α,6α)‐cholestane‐3,6‐diol. Synthesis of the alcohol and subsequent conversion using the Mitsunobu reaction with provide an additional amino-functionalized carbon capable of being incorporated into tetraazamacrocycles.


CONTACT INFORMATION
Email: david.martyn@swosu.edu Office Number: CPP 202-C
Phone Number: 580-774-3114
TEACHING: BASIC COURSES
CHEM 2612 Principles of Lab Safety
CHEM 1203 Gen Chem I
CHEM 1252 Gen Chem I Lab
CHEM 1303 Gen Chem II
CHEM 1352 Gen Chem II Lab
TEACHING: ORGANIC CHEMISTRY
CHEM 3013/3015 Org Chem I
CHEM 3111/3015L Org Chem I Lab
CHEM 4113/4115 Org Chem II
CHEM 4021/4115L Org Chem II Lab
CHEM 4013 Materials Chemistry
OTHER ACTIVITIES
AUG 2021-PRESENT
Chemistry Club Sponsor
RECENT PUBLICATIONS
Palwai, Naveen R.; Martyn, David E.; Neves, Luis F. F.; Tan, Yongqiang; Resasco, Daniel E.; Harrison, Roger G. “Retention of Biological Activity and Near-Infrared Absorbance Upon Adsorption of Horseradish Peroxidase on Single-Walled Carbon Nanotubes” Nanotechnology, 2007, 18, 235601/1-235601/5.
Halterman, Ronald L.; Pan, Xingang; Martyn, David E.; Moore, Jason L.; Long, Andrew T., “Conformational Interconversions in [2]Catenanes Containing a Wide Rigid Bis(p-benzyl)methyl Spacer” Journal of Organic Chemistry, 2007, 72, 6454-6458.
Halterman, Ronald L.; Martyn, David E. “Translational Isomerizations in [2]Catenanes with Unsymmetrically Substituted Resorcinol-Based Tethers” Journal of Organic Chemistry, 2007, 72, 7841-7848.
Halterman, R.L., Martyn, D.E., Pan, X., Ha, D.B., Frow, M., Haessig, K. “Path Selection for Conformational Interconversions in [2]Catenanes” Organic Letters, 2006, 8, 2119-2121.
