Dr. Tim Hubin is a Professor of Inorganic Chemistry in the Chemistry & Physics Department at Southwestern Oklahoma State University (SWOSU). He has been honored as a Bernhardt Professor of Chemistry and as a Henry Dreyfus Teacher-Scholar.
Dr. Hubin has several patents for his chemistry research and is an active recipient of many research grants. He currently holds seven U.S. and European patents in the fields of oxidation catalysts, consumer product bleach compositions, anti-viral transition metal complexes, and MRI contrast agents.
EDUCATION
1994 – B.S. Secondary Science Education, Kansas State University
1994 – B.S. Chemistry, Kansas State University
1999 – Ph.D. Chemistry, University of Kansas
2005 – Began teaching at SWOSU
2019 – 2024– Chair of Chemistry & Physics at SWOSU
2024 – Present – Professor of Chemistry & Physics at SWOSU
RESEARCH
Development and Screening of Cross-Bridged Transition Metal Complexes
Dr. Tim Hubin (Mentor)
Actively accepting research students for pay and credit.
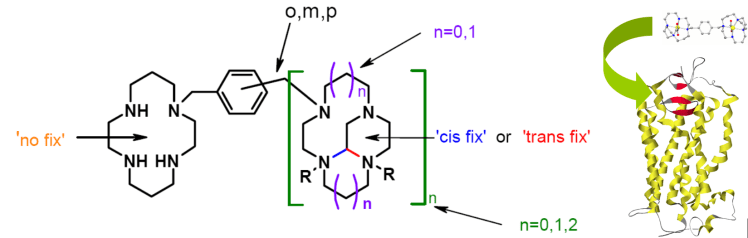
Project 1: CXCR4 Chemokine Receptor Antagonists. CXCR4 chemokine receptors are found on the surface of immune, and other, cells, and together with the specific natural ligand, stromal cell-derived factor-1α (SDF-1α, also known as CXCL12), have been revealed to play a role in a number of disease states. For example, the CXCR4–CXCL12 system has involvement in a number of inflammatory diseases, and the development of rheumatoid arthritis.1,2 Also, within the last ten years the CXCR4 and CCR5 co-receptors have been revealed as the entry route for HIV into cells, generating interest in a new therapeutic approach to treatment via fusion inhibitor drugs rather than the current preference for reverse transcriptase and protease inhibitors.3 CXCR4 expression has also been reported in at least 23 different epithelial, mesenchymal and hematopoietic cancers.4 CXCL12 stimulation of tumor growth, angiogenesis, and metastasis of breast cancer cells has been described.5-7 Target organs for breast metastases such as liver, lung, and bone have high levels of CXCL12, triggering the specific migration of breast tumor cells that express the CXCR4 receptor.8
Specific Aims. Due to the wide-ranging potential biomedical applications that might result, our aim is to develop new antagonists for the CXCR4 co-receptor. They are conformationally fixed macrocyclic compounds where the unrestrained equivalent is a known CXCR4 antagonist. We have successfully demonstrated our concept in the synthesis of several of our proposed compounds. We now intend to complete the synthesis and testing of a series of new compounds, and to learn more about the essential design features for this drug class through spectroscopic and biological studies.
Methods. The project involves the synthesis and characterization of the chemical properties of new analogues in the series of compounds pictured below. Special emphasis would be placed on synthesizing biologically active compounds and on growing crystals for structural studies.
Project 2: Polymeric Transition Metal Oxidation Catalysis for Water Purification Over the last 20 years, we have established a series of transition-metal complexes with rigid, cross-bridged macrocyclic ligands.5-10 These complexes are extremely stabile compared to other, less topologically constrained complexes–the half-life of Cu(Me2EBC)Cl2 in 1 M HClO4 was found to be > 6 years at 313 K, which was eight orders of magnitude slower decomposition than Cu(Me4Cyclam)2+ ligand (t1/2 = 2 seconds), the simplest unbridged macrocyclic analogue.5 In 1 M OH– base, Mn(Me2EBC)Cl2 (Figure 1) can be oxidized by oxygen to the corresponding manganese(III) complex, which is stable in air for months.6 Because of their extreme stability over a wide pH range, this class of complexes provides a unique chance to explore applications in remediating structurally diverse pollutants without pH adjustments. It was found that the manganese complex demonstrated remarkable dye degradation activity over pH range 1-13, and detailed investigations revealed that this manganese complex can play multiple roles in pollutant elimination.1 It is this type of environmentally friendly waste water purification that we aim to explore further of the course of this project. To further enhance the utility of such robust oxidation catalysts, we have produced polymer immobilized analogues that are now heterogeneous and can be removed and recycled during the water purification process. (Figure 2)


CONTACT INFORMATION
Email: tim.hubin@swosu.edu Office Number: CPP 205-C
Phone Number: 580-774-3026
TEACHING: BASIC COURSES
CHEM 1004 Gen Chem
CHEM 1004L Gen Chem Lab
CHEM 1203 Gen Chem I
CHEM 1252 Gen Chem I Lab
CHEM 1303 Gen Chem II
CHEM 1352 Gen Chem II Lab
TEACHING: INORGANIC CHEMISTRY
CHEM 2112 Structure & Bonding
CHEM 2612 Principles of Lab Safety
CHEM 3233 Inorganic Chemistry
CHEM 3211 Inorganic Lab
CHEM 4001-3 Chemistry Research
OTHER ACTIVITIES
GRANT RESEARCH AT SWOSU
EPSCOR Research
EPSCOR Page
JULY 2010 – JUNE 2024
SWOSU OK-LSAMP Coordinator
RECENT PUBLICATIONS
“Mechanistic Insights into Iron-Catalyzed C−H Bond Activation and C−C Coupling” Brewer, S. M.; Schwartz, T. M.; Mekhail, M. A.; Turan, L. S.; Prior, T. J.; Hubin, T. J.; Janesko, B. G.; Green, K. N. Organometallics, 2021, 40, 2467-2477. https://doi.org/10.1021/acs.organomet.1c00211
“An ethylene cross-bridged pentaazamacrocycle and its Cu2+ complex: constrained ligand topology and excellent kinetic stability” Shircliff, A. D; Burke, B. P.; Davilla, D. J.; Burgess, G. E.; Okorocha, F. A.; Shrestha, A.; Allbritton, E. M. A.; Nguyen, P. T.; Lamar, R. L.; Jones, D. G.; Gorbet, M.-J.; Allen, M. B.; Eze, J. I.; Fernandez, A. T; Ramirez, D.; Archibald, S. J.; Prior, T. J.; Krause, J. A.; Oliver, A. G.; Hubin, T. J. Chem. Commun., 2020, 56, 7519-7522. https://doi.org/10.1039/d0cc00919a
“Copper-64 PET imaging of the CXCR4 chemokine receptor using a cross-bridged cyclam bis-tetraazamacrocyclic antagonist” Burke, B.; Miranda, C.; Lee, R.; Renard, I.; Nigam, S.; Clemente, G.; D’huys, T.; Ruest, T.; Domarkas, J.; Thompson, J.; Schols, D.; Hubin, T.; Cawthorne, C.; Archibald, S. J. Nucl. Med. 2019, 61, 123-128. DOI: 10.2967/jnumed.118.218008
“Tetraazamacrocyclic derivatives and their transition metal complexes as antileishmanial leads” Hubin, T. J.*; Walker, A. N.; Davilla, D. J.; Carder Freeman, T. N.; Epley, B. M.; Hasley, T. R.; Amoyaw, P. N. A.; Jain, S.; Archibald, S. J.; Prior, T. J.; Krause, J. A.; Oliver, A. G.; Tekwani, B. L.; Khan, M. O. F. Polyhedron 2019, 163, 42-53. https://doi.org/10.1016/j.poly.2019.02.027
“Acetate as a model for aspartate-based CXCR4 chemokine receptor binding of cobalt and nickel complexes of cross-bridged tetraazamacrocycles” Cain, A.N.; Carder Freeman, T. N.; Roewe, K. D.; Cockriel, D. L.; Hasley, T. R.; Maples, R. D.; Allbritton, E. M. A.; D’Huys, T.; van Loy, T.; Burke, B. P.; Prior, T. J.; Schols, D.; Archibald, S. J.*; Hubin, T. J.* Dalton Trans. 2019, 48, 2785-2801. https://pubs.rsc.org/en/content/articlelanding/2019/dt/c8dt04728f DOI: 10.1039/C8DT04728F
“Increase of direct C-C coupling reaction yield by identifying structural and electronic properties of high-spin iron tetra-azamacrocyclic complexes” Brewer, S. M.; Wilson, K. R.;* Jones, D. G.;* Reinheimer, E. W.; Archibald, S. J.; Prior, T. J.; Ayala, M. A.;* Foster, A. L.;* Hubin, T. J.; Green, K. N. Inorg. Chem. 2018, 57, 8890−8902.
